
Savonia Article Pro: Navigating Value in Digital Health: Factors Shaping Digital Medical Device Value Propositions for Healthcare in the EU
Savonia Article Pro is a collection of multidisciplinary Savonia expertise on various topics.
This work is licensed under CC BY-SA 4.0
Healthcare under pressure – why value and innovation matter more than ever
Europe’s health systems are strained due to demographic changes, increasing chronic conditions, and a shortage of healthcare workers, prompting calls for more efficient resource use (OECD & European Commission 2024). Consequently, the idea of value-based healthcare is gaining attention, but a universally accepted definition of value within this framework does not yet exist. Stakeholders such as patients, physicians, providers, and medical device manufacturers have differing interests, leading to a lack of consensus on what constitutes value. (European Commission 2019, 5-9). Also, healthcare and technology sectors are often viewed as disconnected, which hampers the translation of scientific discoveries into practical medical devices (Bergsland, Elle & Fosse 2014). Nonetheless, innovative digital medical devices present substantial opportunities to increase access to services, support, and replace traditional care, thereby providing added value and convenience to patients (Essén et al. 2022).
Innovation at a crossroads of stakeholder demands and regulatory hurdles
In the European Union (EU), this fast-growing industry – mainly driven by small and medium-sized enterprises – is crucial for providing quality care and acts as a key economic contributor both within Europe and globally (European Commission n.d.). However, digital medical device manufacturers encounter difficulties in demonstrating their value to various stakeholders and in meeting the EU’s regulatory demands. Consequently, many companies, especially in their startup stages, decide to aim their product towards consumers or focus on other markets, such as the United States (Maresova, Rezny, Peter, Hajek & Lefley 2021; Mantovani, Leopaldi, Nighswander & Di Bidino 2023).
Purpose and methods
The master’s thesis “Navigating Value in Digital Health: Factors Shaping Digital Medical Device Value Propositions for Healthcare in the EU” (2026) sought to identify sector-specific factors affecting the value propositions of digital medical devices aimed at EU healthcare systems. The study considered the perspective of small- and medium-sized enterprises seeking to enter the market. It combined a narrative literature review with inductive content analysis, examining ten peer-reviewed articles published between 2024 and 2025.
Findings
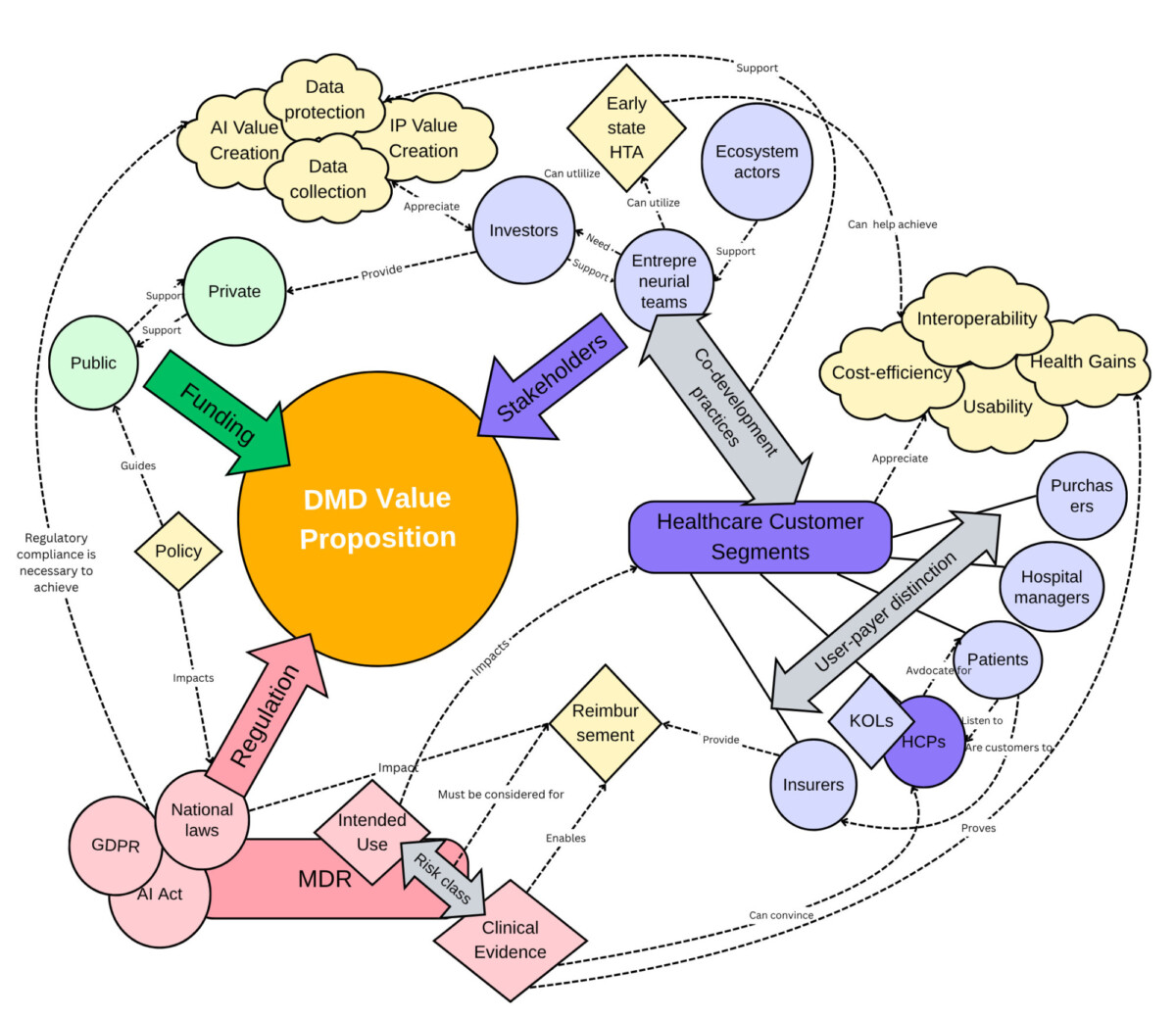
The study revealed that value propositions for digital medical devices targeting EU healthcare systems are shaped by multiple stakeholder perspectives, regulatory frameworks, and funding decisions. These elements are interconnected, influenced by shifting cultural and political trends, and may differ across EU member states. The results indicate that companies developing digital medical devices can enhance their value propositions for healthcare customer segments within the EU by understanding these factors and staying updated on future directions. The research introduced structured tools, such as early-stage health technology assessment, to help navigate this complex environment. The insights can be leveraged not only by medical device ventures but also by anyone aiming to comprehend digital health dynamics in the EU.
Factors shaping digital medical device value propositions for healthcare in the EU
Authors
Meri Mäkelä, Student (Digital Health), Master School, Savonia UAS, Kuopio, Finland
Bryn Lane, CFA, Adjunct Lecturer, Master School, Savonia UAS, Kuopio, Finland
Liisa Klemola, PhD, R.N., Senior Lecturer, Master School, Savonia UAS, Kuopio, Finland
References
Bergsland, J., Elle, OJ. & Fosse, E. 2014. Barriers to medical device innovation. Medical Devices: Evidence and Research 7, 205-209. https://doi.org/10.2147/MDER.S43369. Accessed 16.9.2025.
Essén et al. 2022. Health app policy: international comparison of nine countries’ approaches. npj Digital Medicine 5 (31). https://doi.org/10.1038/s41746-022-00573-1. Accessed 11.3.2026.
European Commission 2019. Directorate-General for Health and Food Safety, Defining value in ‘value-based healthcare’. Report of the Expert Panel on Effective Ways of Investing in Health. Publications Office. https://data.europa.eu/doi/10.2875/35471. Accessed 25.11.2025
European Commission n.d. Public Health – Medical Devices Sector – Overview. Website of the European Union. https://health.ec.europa.eu/medical-devices-sector/overview_en/. Accessed 1.9.2025.
Mantovani, A. Leopaldi, C. Nighswander, C. & Di Bidino, R. 2023. Access and reimbursement pathways for digital health solutions and in vitro diagnostic devices: Current scenario and challenges. Frontiers in Medical Technology 5, 1101476. https://doi.org/10.3389/fmedt.2023.1101476. Accessed 31.5.2025
Maresova, P., Rezny, L., Peter, L., Hajek, L. & Lefley F. 2021. Do Regulatory Changes Seriously Affect the Medical Devices Industry? Evidence From the Czech Republic. Frontiers in Public Health 9, 666453. https://doi.org/10.3389/fpubh.2021.666453. Accessed 11.9.2025.
OECD & European Commission 2024. Health at a Glance: Europe 2024: State of Health in the EU Cycle. OECD Publishing. https://doi.org/10.1787/b3704e14-en. Accessed 25.11.2025.